Encephalitis Viruses Japanese Encephalitis Virus (JE) Western Equine Encephalitis (WEE) Venezuelan Equine Encephalitis (VEE)
advertisement

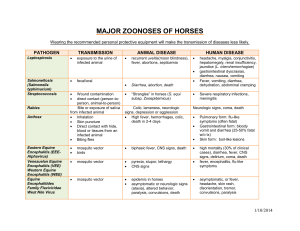
Encephalitis Viruses Japanese Encephalitis Virus (JE) Western Equine Encephalitis (WEE) Venezuelan Equine Encephalitis (VEE) Eastern Equine Encephalitis (EEE) Principal investigators are responsible for communicating this information to staff working with or around this agent, and for mitigation of associated risks. This document is not intended to be used as a sole source for diagnosis, medical treatment, or medical advice. Consult a CSU Authorized Treating Physician for concerns about work related medical conditions. CONTAINMENT AND SPECIAL PRECAUTIONS Containment BSL‐3 Level practices, containment equipment and facilities are required for work involving potentially infected materials, animals, cultures, or mosquitos. Special considerations: North American strains of EEE virus and some epizootic subtypes (IAB and IC) of VEE virus are Select Agents Arthropod‐borne disease Can cross placenta HAZARD IDENTIFICATION Disease: Encephalomyelitis Transmission: infected mosquitoes, aerosol transmission of VEE and WEE viruses, natural person to person spread not reported, no human to mosquito transmission for WEE and EEE virus, but can happen in VEE virus up to 72 hours post‐ infection, VEE virus known to cross the placenta and this may also occur with the other viruses. Incubation: 1‐6 days (VEE) 5‐15 days (JE, WEE and EEE) Infectious dose: VEE – 1 pfu, JE, WEE and EEE – unknown VIABILITY/INACTIVATION Stability: Stable in blood, exudates, and freeze dried materials (VEE), can survive over winter in mosquito eggs (JEE) Chemical Inactivation: Like most enveloped viruses, susceptible to 1% bleach (500 ppm available sodium hypochlorite), 2% glutaraldehyde, 3‐8% Formaldehyde, quaternary compounds and phenolics. JEE and VEE are susceptible to 70% ethanol. EEE is inactivated after 60 minutes exposure to 50% ethanol. Physical Inactivation: Sensitive to autoclave and drying Page 1 of 4 Colorado State University Environmental Health Services Biosafety Office Updated 2013 (970) 491‐0270 **Disclaimer** This document is for informational purposes ONLY. This document should not be used in lieu of professional medical attention, and medical professionals should seek appropriate resources for diagnosis and treatment.** MEDICAL Signs and symptoms: EASTERN EQUINE ENCEPHALITIS . Fever Chills Myalgia (muscle pain) Arthalgia (joint pain) Headache Irritability Neck stiffness Confusion Stupor Disorientation Tremors Seizures Paralysis Coma Abdominal pain Vomiting and diarrhea Symptoms subside in 1‐2 weeks JAPANESE ENCEPHALITIS . Fever Headache Stupor Disorientation Coma Tremors/Seizures Paralysis Diarrhea Myalgia (muscle pain) WESTERN EQUINE ENCEPHALITIS (similar signs as EASTERN EQUINE ENCEPHALITIS) . Fever Chills Myalgia (muscle pain); back pain Malaise (discomfort) headache Nausea, vomiting Diarrhea, abdominal pain Respiratory symptoms Symptoms subside in 1‐2 weeks VENEZUELAN EQUINE ENCEPHALITIS . Fever Chills Malaise (discomfort) Myalgia (muscle pain) Severe headache Encephalitis Coughing Sore throat Nausea, vomiting Diarrhea Symptoms subside in 4‐6 days Pre‐exposure prophylaxis: JE: Vaccine readily available, although there are no data demonstrating vaccine efficacy post needle stick or aerosol exposure. EEV, VEE, and WEE‐ May be available under certain circumstances through USAMRIID Diagnosis: In all cases, Serum is taken on day of exposure, and 10‐14 days post infection to detect 4‐fold rise in titer. EEE: Isolated in A549 and MRC‐5 cell cultures. Antigens detected by immunofluorescence and ELISA. Nucleic acid detected by RT‐PCR. WEE: Throat swabs can be cultured. Viral isolation in embryonated eggs (Vero cell plaque assay). Also, detection methods similar to EEE. Page 2 of 4 Colorado State University Environmental Health Services Biosafety Office Updated 2013 (970) 491‐0270 **Disclaimer** This document is for informational purposes ONLY. This document should not be used in lieu of professional medical attention, and medical professionals should seek appropriate resources for diagnosis and treatment.** VEE: Viral isolation from blood, CSF and throat swabs. During febrile stage, antigen capture ELISA can detect VEE in the blood. Also, detection methods similar to EEE and WEE. JE: Similar to EEE, WEE and VEE Treatment (Post‐Exposure Prophylaxis/Treatment): Treatment is supportive and symptomatic WHAT TO DO IF AN EXPOSURE OCCURS Employees, Graduate Students, Work Study 1. Employee notifies Biosafety (970‐491‐0270) and/or Occupational Health Program Coordinator (970‐420‐8172) to inform where medical attention will be sought and if transportation is needed The Principal Investigator/Supervisor must also be notified 2. Employee goes to Emergency Room 3. After the Emergency Room visit, individual fills out the following forms: Biosafety Incident report form: http://www.ehs.colostate.edu/WBiosafety/PDF/IncidentReportForm.pdf Workers’ Compensation (within 4 days or as soon as possible): http://www.ehs.colostate.edu/WWorkComp/Home.aspx 4. Employee follows up with CSU Authorized Treating Physician Student Not Paid by CSU 1. Contact supervisor/PI 2. Student or supervisor contact Biosafety (491‐0270) or Occupational Health (420‐8172) to inform where attention is being sought, and to arrange transportation if needed 3. Student goes to CSU Health Network (formerly Hartshorn Health Services) 4. After the visit to CSU Health Network, student fills out Biosafety Incident Report form http://www.ehs.colostate.edu/WBiosafety/PDF/IncidentReportForm.pdf Volunteers and Visitors 1. Contact supervisor/PI 2. Contact Biosafety (491‐0270) or Occupational Health (420‐8172) to inform where attention is being sought, and to arrange transportation if needed 3. Individual goes to their personal physician, or as otherwise directed by their physician 4. Individual fills out Biosafety Incident Report form http://www.ehs.colostate.edu/WBiosafety/PDF/IncidentReportForm.pdf REFERENCES Bell M. Exotic viral diseases: a global guide. By Berger SA, Calisher CH, Keystone JS. Emerg Infect Dis 2003; 9:1357. (http://www.cdc.gov/ncidod/EID/vol9no10/03‐0490.htm CDC General Information: http://www.cdc.gov/ncidod/dvbid/jencephalitis/qa.htm Iowa State University Technical Fact Sheet, Eastern, Western, Venezuelan: http://www.cfsph.iastate.edu/Factsheets/pdfs/easter_wester_venezuelan_equine_encephalomyelitis.pdf Iowa State University Technical Fact Sheet, Japanese: http://www.cfsph.iastate.edu/Factsheets/pdfs/japanese_encephalitis.pdf Page 3 of 4 Colorado State University Environmental Health Services Biosafety Office Updated 2013 (970) 491‐0270 **Disclaimer** This document is for informational purposes ONLY. This document should not be used in lieu of professional medical attention, and medical professionals should seek appropriate resources for diagnosis and treatment.** Public Health Agency of Canada, Equine Encephalitis: http://www.phac‐aspc.gc.ca/lab‐bio/res/psds‐ftss/equine‐ eng.php Public Health Agency of Canada, Japanese Encephalitis: http://www.phac‐aspc.gc.ca/lab‐bio/res/psds‐ftss/msds173e‐ eng.php Public Health Agency of Canada, Venezuelan Equine Encephalitis: http://www.phac‐aspc.gc.ca/lab‐bio/res/psds‐ ftss/ven‐encephalit‐eng.php CONTENT REVIEW This document has been reviewed by: CSU subject matter experts: Dr. Richard Bowen Page 4 of 4 Colorado State University Environmental Health Services Biosafety Office Updated 2013 (970) 491‐0270 **Disclaimer** This document is for informational purposes ONLY. This document should not be used in lieu of professional medical attention, and medical professionals should seek appropriate resources for diagnosis and treatment.**